Uniqueness of DNA
Our unique, individual genetic make-up has a great impact on who we are. Across mankind, every single day millions of tiny mutations and variations happen within the genomes that make up our DNA. Together with our age, environment and lifestyle choices, these inborn and acquired variations make a huge difference in how we break down caffeine or respond to medication. And no matter how much we know about our genetic makeup, we’re always at risk of accidents and illnesses.
From tearing a meniscus while playing tennis to developing colon cancer, the longer we live, the more likely we are to need medical treatment. But with no two people being the same – not even identical twins share the exact same DNA – wouldn’t it be good if we could predict how people will respond to treatment ahead of time? If we could grow new organs in the laboratory rather than having patients wait on a transplant list?
Regenerative Medicine
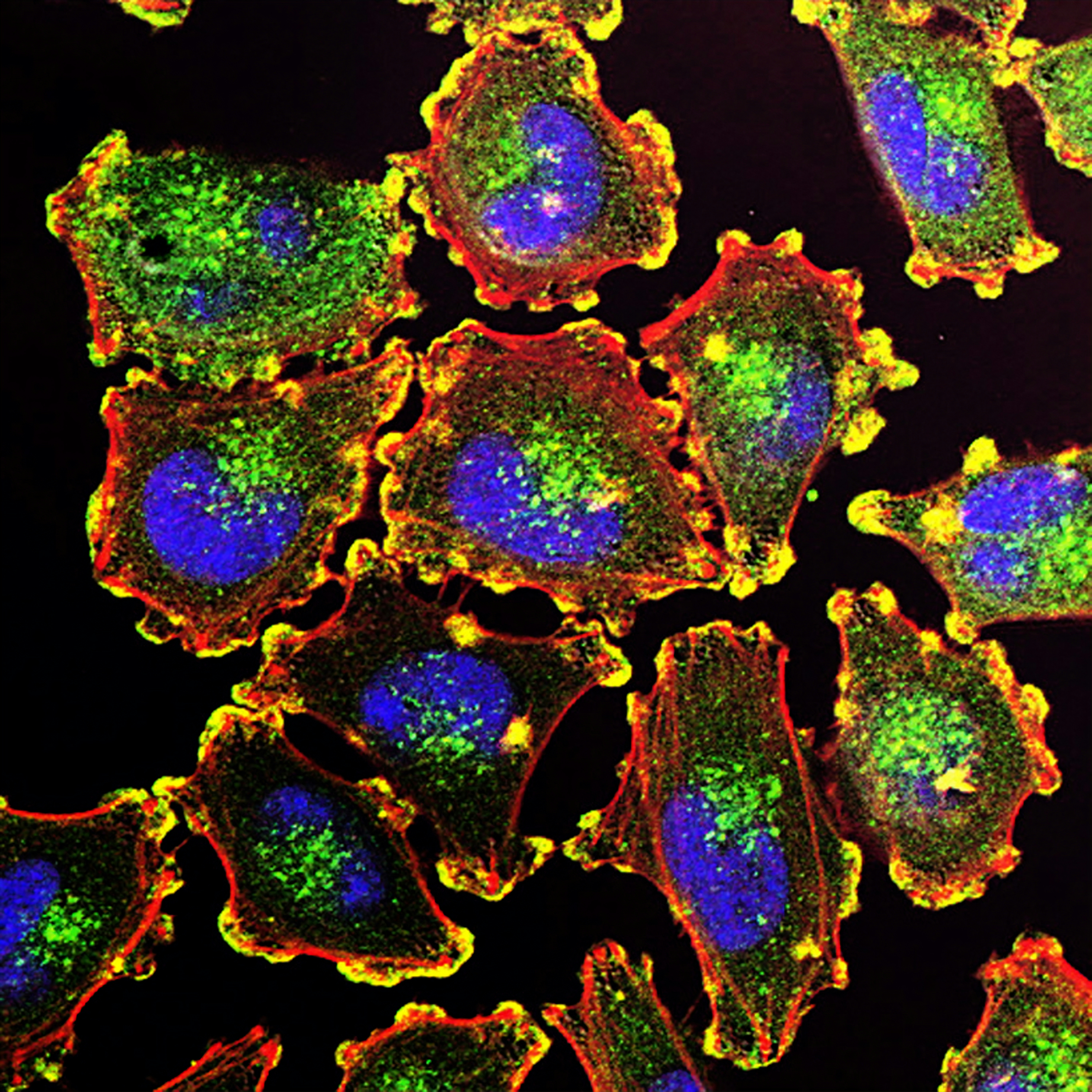
Stem cells are one of Mother Nature’s greatest gifts. Able to restore function after tissue has been damaged, nothing in the natural world can rival the regenerative power of a stem cell. They are the very source of life. Embryonic stem cells can become any type of cell in our body, and adult stem cells can mature into the cell types of the tissue they originate from.
And with much of today’s treatments working on a cellular level, it’s hardly surprising that biomedical research teams at the University Medical Centre (UMC) in Utrecht are focussed on unravelling the underlying scientific principles behind stem cell biology, biofabrication and gene editing. With their work, the scientists at UMC aim to transform modern medicine with better diagnostics, more effective therapies, and quicker healthcare decisions.
Mini Organs
Back in 2009, Utrecht based stem cell biologist Hans Clevers made a discovery that would change stem cell research for good. Within his lab, researchers had used mouse intestinal stem cells to grow something in a petri dish that looked like the real intestinal tissue in the mouse. In 2011, they showed a similar concept for human intestinal stem cells. It was a first patient-derived organoid, or mini organ, and revolutionised how we can study human tissue in the lab, with great potential for drug discovery and personalized medicine.
Today, teams at UMC are using liver organoids and bioprinting to create a reliable model system that can predict drug-induced liver injury before a drug enters the pre-clinical phase. Drug-induced liver injury is the leading cause of acute liver failure in the developed world and one of the main reasons for lack of drug efficacy during clinical trials. As the organoids are of human origin, they can predict toxicity more reliably.
(Almost) Limitless Power
Similarly, normal and tumour organoids are being used to modulate newly identified regulators, such as Wnt proteins, and screen new drugs. Wnt proteins are tiny molecules that are involved in everything from organ development to tissue regeneration and homeostasis. The biggest challenge in designing new molecules? Ensuring they don’t upset other essential biological processes.
“Human stem cells can really transform how we develop and use medication in an individual setting,” says prof. dr. Jeffrey Beekman, principal investigator at UMC. “By studying patient-derived stem cell models such as organoids in the lab, we can very precisely quantify individual biology in the context of disease or therapy. In this way, we have worked out how a genetic disease such as cystic fibrosis can be measured in the lab in organoids, and also how the disease can be repaired with medications on an individual basis. As the stem cells can grow indefinitely, we can create large quantities of cells and study more than 1000 medications per patient , all in the lab without bothering the patient. Our cystic fibrosis test was a first clinical application of organoids and is currently used worldwide to study who can benefit from new cystic fibrosis drugs.”
Bioengineering Medical Breakthroughs
Once considered the garbage bins of a cell, extracellular vesicles, or EVs, carry debris out of cells. We now know that they help cells communicate with each other, with one cell being able to receive EV from another cell. Researchers are now using stem cell derived EVs to transport factors that are essential for tissue repair and regeneration, for example in a damaged heart. The UMC team is currently designing a clinical grade, GMP compatible bioreactor for the scalable isolation and production of EVs.
Even more, the team is also piloting a clinical trial in which mesenchymal stem cells (MSCs) are injected into the major muscles in the hands and arms of patients with systemic sclerosis, a debilitating autoimmune disease. Other diseases currently under the stem cell microscope include facioscapulohumeral muscular dystrophy – one of the most frequently inherited muscle wasting diseases and one for which there is no cure or effective treatment –
From finding the correct treatment for genetic diseases, infectious diseases and different types of cancer, to better understanding neurological illnesses such as schizophrenia, bipolar disorder and autism, the ways in which stem cells and organoid based research can improve healthcare are limitless. The hope is that eventually a wide variety of diseases can be treated either through cellular therapy or by stimulating the body’s own stem cells into repairing their own tissues.